Clinical geneticist and associate professor Stefan Barakat began a new project 2.5 years ago: to map the DNA of a hundred patients who had failed to receive a diagnosis. This was possible because whole genome sequencing, a technique for mapping the entire DNA, has become cheaper and more accessible in recent years.
The first two patients were an instant hit. The researchers found a mutation in the DNA of two teenage brothers that was previously overlooked in routine diagnostics. The brothers had been patients at the Erasmus MC Sophia Children’s Hospital for several years because of an unknown syndrome. Walking was difficult for them, mainly because the upper leg muscle tightened too much. A diagnosis remained elusive. Until Barakat and his team found a mutation on both copies of the AMFR gene.
Stem cells
The function of the AMFR gene was still unknown at that time. Barakat: ‘There was virtually no literature to be found, so we had to do our own investigation into this gene. The team took brain cells fabricated from stem cells and inserted a mutation that turned off the AMFR gene. The brain cells then showed increased cholesterol storage.
If the endoplasmic reticulum is the maintenance shop, this gene is one of the janitors.
The AMFR gene appears to act on the endoplasmic reticulum, which Barakat likes to call the maintenance shop of the cell. ‘If the endoplasmic reticulum is the maintenance shop, this gene is one of the janitors. It regulates proteins that both stimulate and restrict fat production. If the AMFR gene is not working properly, that ratio gets out of balance, and you get an excess of cholesterol, especially in nerve cells.’

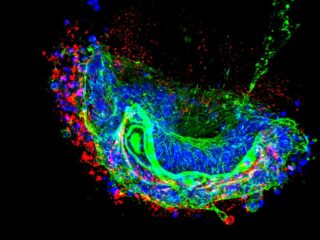
Cholesterol (in red) accumulates in a cell. On the left is a normal cell, and on the right with a mutation in the AMFR gene.
Most people know cholesterol as fat they must avoid to prevent heart problems. But cholesterol is also present in cells as a building block, for example, in the cell membrane: the cell’s outer layer. If there is too much cholesterol, the membrane becomes stiff; if there is too little, it becomes flaccid. ‘Some nerve cells transmit signals from your brain to your legs and can be as long as a meter and a half. You can imagine that if the lipids are out of balance in cells like that, signals can no longer get through properly,’ Barakat says. That may explain why the brothers had trouble walking.
Swimming ability
On to the next step: the researchers insert the same AMFR mutation into the zebrafish larvae. By doing so, they confirm Barakat’s theory. These fish have a similar motor disorder as the patients; their swimming ability is not as good. And the larvae’s nerve cells appear to be much less branched out.
The researchers do not have to search long for a possible drug. They already know these problems arise due to cholesterol metabolism. Many people already have cholesterol-lowering medications in their medicine cabinet. Barakat: ‘I estimate that ten per cent of people in the Netherlands take medication to lower cholesterol.’ When the researchers give the larvae a standard cholesterol-lowering drug during their development, most regain their swimming abilities and develop normal nerve cells.
Safe to use
So now, prescribe the drug to the brothers? It’s not that easy; human children are not zebrafish, nor do they generally take cholesterol-lowering medication. The researchers must first determine whether cholesterol-lowering drugs are as effective in humans. Previous research does show that this drug is safe to use for children, because some already take it for hereditary forms of excessive cholesterol.
Currently, Barakat is talking with colleagues in pediatric neurology to see if they can set up a clinical trial. ‘We are brainstorming about using the Children’s Brain Lab of the Erasmus MC Sophia Children’s Hospital. They have a treadmill and other sophisticated measuring equipment to fully map all the cognitive and motor functions of the teens.’
Once the Medical Ethics Review Board approves their research protocol and the researchers have collected the necessary data, Barakat and his colleagues hope to offer the medication to the brothers. ‘Should the medication catch on, we can monitor the progress precisely to conclude whether the drugs also work in humans. Despite our promising results from the zebrafish, we have a long road ahead.’

Researcher Eva Medico Salsench looks at the zebrafish larvae under the microscope.
Tinder
That means a possible treatment not only for the Rotterdam brothers. On the “Tinder for geneticists” – or the website GeneMatcher – the researchers found 18 other patients worldwide with the same mutation. Chances are that many more patients worldwide with this condition, currently undiagnosed, are only now coming to light because of this study.
Relief
The researchers published their findings in Acta Neuropathologica. And Barakat feels that everything is coming together in this one research: ‘Finding the mutation with new technology, figuring out how the gene works and detecting a possible drug. We discovered something about cholesterol metabolism in brain cells, contributing to fundamental knowledge about the human body. At the same time, the impact is also evident. Not only for the patients, but also for siblings who may be carriers of the genetic predisposition, and for parents who often walk around with misplaced guilt. A diagnosis can already be quite the relief.’
Start your research career at Erasmus MC. Learn more about our job opportunities.